Search
Coronavirus-2019 (COVID-19) vaccination in Australia commenced in February 2021. The first vaccines recommended for use were AZD1222 and BNT162b2, both delivered as a two-dose primary schedule. In the absence of sustained immunity following immunisation, recommendations for booster vaccination have followed. It is likely that periodic boosting will be necessary for at least some Australians, but it is unknown what the optimal booster vaccines and schedules are or for whom vaccination should be recommended.
The Kids is investigating whether a cystic fibrosis medication may be the answer to ear infections, reducing the need for antibiotics and surgery.
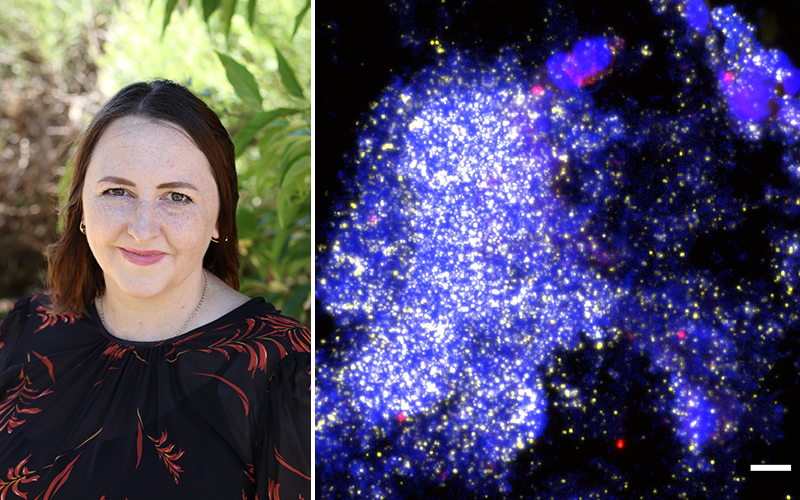
Researchers using powerful microscopes have identified bacterial slime in the lungs of some children with persistent wet coughs.
PICOBOO is a randomised, adaptive trial evaluating the immunogenicity, reactogenicity, and safety of COVID-19 booster strategies. Here, we present data for second boosters among individuals aged 18-<50 and 50-<70 years old primed with BNT162b2 until Day (D) 84.
Invasive pneumococcal disease remains a major cause of hospitalization and death in Papua New Guinean (PNG) children. We assessed mucosal IgA and IgG responses in PNG infants vaccinated with pneumococcal conjugate vaccine (PCV) followed by a pneumococcal polysaccharide vaccine (PPV) booster.
Chris Jennifer Lea-Ann Peter Ruth Brennan-Jones Kent Kirkham Richmond Thornton PhD RN PhD MBBS MRCP(UK) FRACP PhD Head, Ear and Hearing Health
Peter Lea-Ann Ruth Richmond Kirkham Thornton MBBS MRCP(UK) FRACP PhD PhD Head, Vaccine Trials Group Co-Head, Bacterial Respiratory Infectious Disease
Janessa Lea-Ann Peter Ruth Pickering Kirkham Richmond Thornton BSc PhD PhD MBBS MRCP(UK) FRACP PhD Senior Research Fellow (currently HOT NORTH Early
Elke Lea-Ann Ruth Peter Seppanen Kirkham Thornton Richmond BSc PhD PhD PhD MBBS MRCP(UK) FRACP Program Manager, Bacterial Respiratory Infectious
Ruth Peter Thornton Richmond PhD MBBS MRCP(UK) FRACP Co-head, Bacterial Respiratory Infectious Disease Group (BRIDG) Head, Vaccine Trials Group
